2021-01-30 1394

To quote this article:
Chen Wei, Liu Mimi, Liang Yuanbo, Qu Jia*. Real-world research practice of Boao Lecheng ophthalmology licensed medical devices and clinicians' thinking [J]. China Food and Drug Administration. 2020.11 (202): 32-35.
Real-world research practice of Boao Lecheng ophthalmology licensed medical devices and thinking of clinicians
The Practice of Real World Research of Ophthalmic Licensed Medical Devices in Boao Lecheng and Reflection of Clinicians
Chen Wei
Affiliated Eye Hospital of Wenzhou Medical University
Boao Super Hospital International Optometry Center
National Eye, Ear, Nose and Throat Disease Clinical Research Center
CHEN Wei
Eye Hospital of Wenzhou Medical University
International Eye Center, Boao Super Hospital
National Clinical Research Center for Eye and ENT Diseases
Liu Mimi
Boao Super Hospital International Optometry Center
LIU Mi-mi
International Eye Center, Boao Super Hospital
Liang Yuanbo
Affiliated Eye Hospital of Wenzhou Medical University
National Eye, Ear, Nose and Throat Disease Clinical Research Center
LIANG Yuan-bo
Eye Hospital of Wenzhou Medical University
National Clinical Research Center for Eye and ENT Diseases
Qu Jia
Affiliated Eye Hospital of Wenzhou Medical University
Boao Super Hospital International Optometry Center
National Eye, Ear, Nose and Throat Disease Clinical Research Center
QU Jia
Eye Hospital of Wenzhou Medical University
International Eye Center, Boao Super Hospital
National Clinical Research Center for Eye and ENT Diseases
Abstract
As the first batch of specialist teams to settle in Boao Super Hospital, the Department of Ophthalmology has carried out 12 licensed introduction projects, including Boston artificial cornea, Oconvision YUTIQ and scleral lenses, based on the State Council's double "National Nine-Article" licensed medicine and equipment policy. Among them, the glaucoma minimally invasive drainage tube XEN. project was officially approved by the State Food and Drug Administration on March 26, 2020, becoming the first licensed product to be registered and marketed using clinical real-world data. In order to further promote the exploratory practice of using real-world data and evidence at home and abroad to speed up the review of innovative drugs and devices, this paper summarizes and considers the real-world data research work of ophthalmology in the Boao Lecheng International Medical Tourism Pilot Zone, in order to provide clinical real-world data research. refer to.
Based on the "national nine articles" issued by the State Department, the ophthalmology team, as one of the first specialized teams to be stationed in Boao super hospital, has carried out 12 licensed import projects including Boston artificial cornea, YUTIQ and scleral lens, etc. XEN gel, a minimally invasive drainage tube for glaucoma, was officially approved by the State Direct Drug Administration on March 26, 2020 and became the first licensed product by applying clinical real world data. In order to promote the exploratory practice of using real world data and evidence to speed up the evaluation of innovative medicine and devices, the current study summarized and explored the researches of ophthalmic real world data in Boao Lecheng International Medical Tourism pilot area.It may provide information for future clinical real world data research in China.
KeywordsKey words
Licensed medicinal devices; clinical real-world data; Boao Lecheng Pilot Zone; review of innovative medicinal devices; product launch
licensed medicine applicator; clinical real world data; Boao Lecheng pilot area; innovative medicine applicator evaluation; product launch

On January 7, 2020, the State Food and Drug Administration issued the "Guidelines for Real-World Evidence Supporting Drug Development and Review (Trial)" [1]. This is a milestone in the development and review of drugs in my country. On the one hand, it can speed up the review of medical devices in my country, so that innovative technologies and products can benefit the people as soon as possible. On the other hand, normative guidance documents can also be provided for the conduct of real-world research. However, there is still a lot to be explored in the practical process of how real-world research supports drug development and review. The pilot experience of Boao Lecheng's real-world data research and the advanced platform and system formed by relying on innovative technologies will bring new opportunities for my country's larger-scale and higher-level clinical research.
01Practice of real-world research on Boao Lecheng licensed ophthalmic medical devices
The International Optometry and Ophthalmology Center of Boao Super Hospital (hereinafter referred to as the Center) is established by the team of Professor Qu Jia from the National Eye, Ear, Nose and Throat Disease Clinical Research Center, the Affiliated Eye and Optometry Hospital of Wenzhou Medical University and the Zhejiang Eye Hospital. The center settled in Boao Super Hospital on March 31, 2018 with the "1+X" innovative model. Relying on the advantages of large demand for ophthalmology, relatively independent disciplines, good safety, and ease of introduction and development, the center will focus on the introduction of licensed medicines. Twelve licensed projects have been introduced so far. Among them, Allergan's XEN. glaucoma drainage tube and Johnson & Johnson's Catalys femtosecond laser system were selected for the first batch of clinical real-world data research pilot projects of the State Food and Drug Administration. On March 26, 2020, the State Food and Drug Administration approved the registration of Allergan's "glaucoma drainage tube" [2]. This product is the first product in the pilot program that uses the clinical real-world data collected in the Hainan Boao Lecheng Pioneer Zone combined with overseas real-world evidence to evaluate ethnic differences. It is also the first product approved for registration in China through this method. .
At present, there are 5 ophthalmology candidates that are listed as the second batch of drug and device candidates for registration applications through real-world data research.
02Characteristics and experience of real-world research on Boao Lecheng licensed ophthalmic medical devices
After the glaucoma minimally invasive drainage tube product was approved for marketing, real-world data research has received extensive attention from related industries. As the research subject of real-world research and the collector of clinical real-world data, in the specific implementation process, the author summarizes its characteristics and experience that are different from other clinical research.
Boao Lecheng is currently the only area in the country that can use licensed pharmaceutical products that have been listed abroad and not listed in China [3-7]. Patients with licensed medical needs from all over the country are the first people in my country to receive international innovative drug and device treatment. Patients come from all over the country, there are many physicians participating in multi-site clinical diagnosis and treatment in the ophthalmology center, the implementation experience of intervention measures may affect the treatment results, and the standardization of follow-up data collection in different hospitals after treatment is completed. Factors of research quality in real-world data. The author's experience is: after the project is launched, clinical experts, research plan design and statistics experts, and pharmaceutical R&D contract outsourcing service agencies (contract research organization, CRO) should be organized as soon as possible to form a multidisciplinary team to carry out work; plan training should be strengthened before the plan is launched; The surgical training and qualification shall be uniformly trained and accredited by internationally renowned experts invited by the sponsor.
The Boao Lecheng licensed ophthalmic medical device real-world data research project involves the participation of many other domestic ophthalmic centers. The intervention implementation center is unified in the Boao Super Hospital, which is in line with the traditional multi-center clinical in terms of ethical approval, data quality responsibility, medical qualifications, etc. Research is quite different. The author's experience is: each ophthalmology center participates in the researcher as an individual, the lead researcher signs a participation agreement with the individual researcher, and the ethics are reviewed by the ethics committee of the Boao Super Hospital [8].
Real-world research integrates research into clinical practice, and is the most popular research by physicians in a real medical environment, but the requirements for rigor, integrity, and traceability of research records are much higher than those of clinical medical records [9]. This creates a huge extra workload for doctors and paramedics. Although some project specialists are hired, this is still a big challenge to carry out the Boao real-world research project, and doctors (researchers) need to carry out heavy research form records in addition to the intensive implementation of innovative technologies.

03Thinking about the real-world research of Boao Lecheng licensed ophthalmic medical devices
The state supports the Boao Lecheng International Medical Tourism Pilot Zone to promote the review and approval of imported drugs and devices through real-world evidence, while scientific supervision requires real-world data research that generates real-world evidence to meet the following conditions: ① Whether the research is conducted in compliance with regulations and ethics Regulatory requirements; ② Can the study design provide reliable scientific evidence to answer the corresponding regulatory questions; ③ Can clinical real-world data meet the applicability standards of data reliability and relevance. This will bring opportunities for policy, system, system and platform innovation for clinical real-world data research.
In my country, real-world research has unique advantages. my country is rich in clinical case resources, and clinicians are efficient in diagnosis and treatment, and a large number of clinical research samples can be obtained in a short period of time to form big data. However, traditional clinical research often requires the establishment of a dedicated clinical public platform and clinical data collection team [10]. Due to the lack of standardized data collection, management process and structured clinical information, the rich clinical resources of hospitals in my country have not become real big data. "Good Clinical Practice for Drugs" (GCP) promotes the development of clinical research in my country, especially randomized controlled clinical trials. The development and exploration of rigorous and standardized real-world research for the purpose of product registration will surely accelerate the standardization of real-world research in my country, and promote the "data" of my country's clinical case resources and the "scientific research" of clinical diagnosis and treatment processes, and improve my country's clinical diagnosis and treatment. level.
One of the biggest barriers to real-world research is the incompleteness and non-shareability of data. Relying on new information innovation technology, realizing the informatization of clinical data collection mode is an important way and means to overcome this obstacle. It is necessary to upgrade the electronic medical record system based on clinical medical management, and treat medical records as data; at the same time, it is necessary to integrate electronic data acquisition system, electronic follow-up system and wearable electronic equipment. Open up the connection between the hospital scientific research platform and the out-of-hospital health care big data platform to realize data sharing between different hospitals and institutions. These all require further policy and institutional innovation.
The current clinical workload of Chinese doctors is often several times that of foreign doctors of the same seniority. To carry out research work without increasing the burden on clinicians, the research efficiency of doctors should be improved and the time spent by doctors in collecting clinical data should be reduced. The acceptability of voice recognition, video recording, and audio recording of data collection, as well as the sorting of audio recordings, require artificial intelligence assistance.
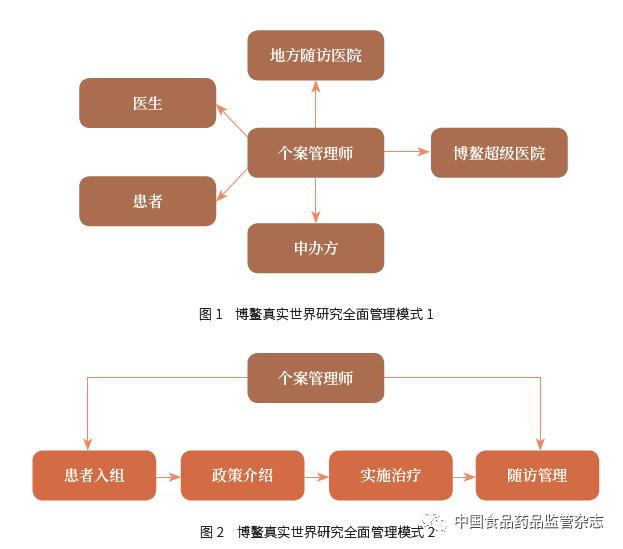
The separation of treatment and follow-up in Boao real-world research may be a norm for future medical models. With the establishment of the integration of the national medical insurance system, free medical treatment and mobile medical treatment (that is, medical treatment in one place and follow-up in other places) may become a norm, which requires changing the mode of disease diagnosis and treatment. Centering on the hospital, extending to the pre-hospital, in-hospital and post-hospital areas, it has become a medical service model centered on disease management to better carry out real-world research. The Eye Hospital Affiliated to Wenzhou Medical University established a case management model for ophthalmic diseases as early as 2015, as shown in Figures 1 and 2. The case manager is a senior nurse who implements a comprehensive follow-up management of the patient. Under the case management model, the follow-up rate of glaucoma in the Eye and Optometry Hospital Affiliated to Wenzhou Medical University can reach 80% [11].
04Summary and Outlook
While serving the health of the people in the Boao Lecheng Pioneer Zone, the innovative ophthalmic medical devices have accumulated preliminary experience for the development of real-world ophthalmology research in China, and also put forward more requirements for the large-scale development of real-world research in my country in the future, such as high efficiency and precision. The establishment of the information-based data collection platform, the integration of the hospital electronic medical record system and the electronic data collection system, as well as intelligent speech recognition, image data recording, etc. The innovative integration of innovative technologies, policies and systems in the Lecheng clinical real-world data application pilot study will provide a good development opportunity and impetus for a wider range of real-world research and clinical research in China.
About the first author
Chen Wei, Ph.D., Director of the International Optometry Center of Boao Super Hospital and Vice President of Wenzhou Medical University Affiliated Ophthalmology Hospital, Professor, Doctoral Supervisor. Specialties: Corneal and Ocular Surface Diseases
About the corresponding author
Qu Jia, Ph.D., Director of the Department of Ophthalmology and Optometry of Wenzhou Medical University, Director of the State Key Laboratory of Ophthalmology and Vision Science jointly built by the provincial and ministerial departments, Director of the National Optometry Engineering Technology Center, professor, doctoral tutor. Specialties: Ophthalmology and Vision Science, Hospital Management
