2021-09-10 1507
Recently, the China Center for Evidence-Based Medicine, West China Hospital of Sichuan University / Hainan Key Laboratory of Real-World Data Research and Evaluation in the Frontier Field of Pharmacology of the State Food and Drug Administration (IF=5.81, JCR District 1) published a report entitled "Assessment" of Traditional Chinese Medicine Interventions Clinical outcomes: Beyond randomised controlled trials" report.
This paper breaks through the traditional thinking of evaluating the efficacy of traditional Chinese medicine based on traditional experiments, and establishes an integrated innovation model of traditional experiments + real-world data research. Medical practice provides a degree of methodological technical preparation.

This paper is jointly led by Professor Sun Xin and Professor Shang Hongcai, the Chinese Evidence-Based Medicine Center of West China Hospital of Sichuan University, the Department of Integrated Traditional Chinese and Western Medicine of West China Hospital of Sichuan University, and the Key Laboratory of Chinese Medicine and Internal Medicine of the Ministry of Education of Beijing University of Traditional Chinese Medicine (Dongzhimen Hospital). Relevant experts work together to complete. It is reported that Frontiers in Pharmacology is one of the most important journals in the field of traditional Chinese medicine efficacy evaluation.
The revitalization of traditional Chinese medicine has become a national strategy. Clinical efficacy is the foundation of the survival and development of traditional Chinese medicine. However, due to the obvious differences between the theoretical basis and logical thinking of traditional Chinese medicine and modern medicine, the evaluation of clinical efficacy of traditional Chinese medicine faces important methodological challenges. Combining the theory and characteristics of traditional Chinese medicine, this article innovatively proposes a problem-oriented "4R" model (eRCT-pRCT-RCDOS-REGOS) for the clinical efficacy evaluation of traditional Chinese medicine in the context of integrated traditional Chinese and western medicine, and applies this method to To solve the evaluation of the efficacy of different proprietary Chinese medicines.
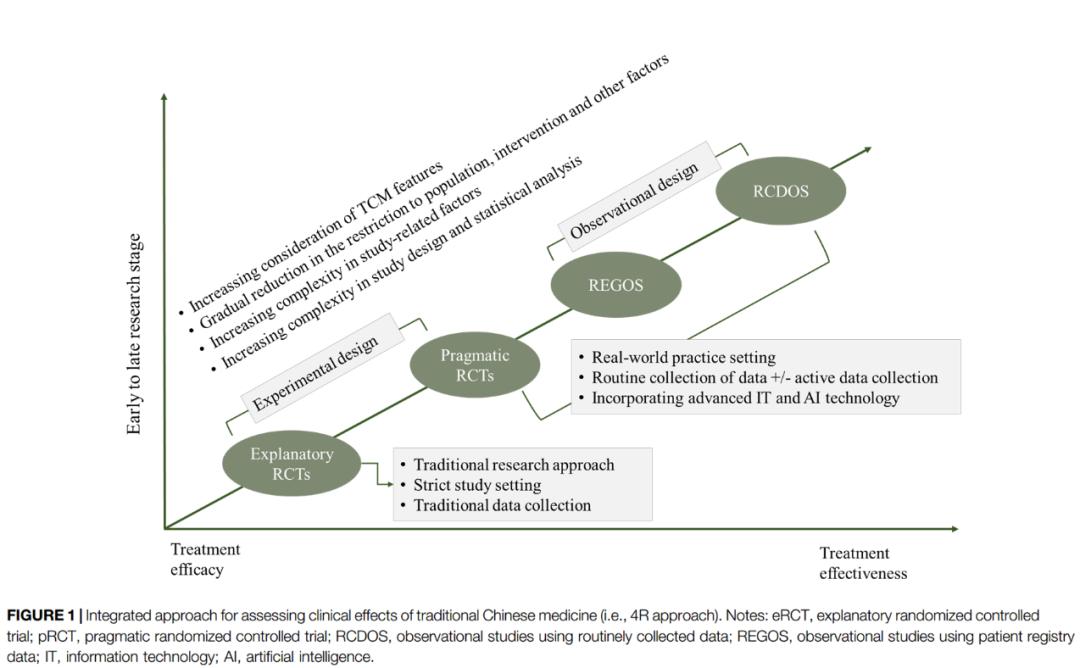
This methodological model helps to systematically support the evaluation of the efficacy of traditional Chinese medicine under the background of integrated traditional Chinese and Western medicine, realize the further integration of clinical research methods and the practice mode of traditional Chinese medicine, and promote the innovation and development of the evaluation model of traditional Chinese medicine.
We look forward to working with domestic colleagues to jointly build an integrated method system for traditional clinical trials and real-world data research, carry out clinical research on traditional Chinese medicine, explore and prove the advantages and characteristics of traditional Chinese medicine in disease prevention and treatment, and promote the inheritance and innovation of traditional Chinese medicine. Promote the modernization and internationalization of traditional Chinese medicine.
About Hainan Key Laboratory of Real World Data Research and Evaluation
Hainan Key Laboratory of Real-world Data Research and Evaluation is the first key laboratory of real-world data research and evaluation in China. This key laboratory consists of three units: West China Hospital of Sichuan University, Boao Super Hospital, and Hainan Boao Pilot Area Adverse Drug Reaction Monitoring Center. Build, manage and share together. Relying on the national strategy of Hainan Free Trade Port construction, the laboratory is guided by the innovation of the national drug and device supervision system and mechanism, integrating the comprehensive clinical advantages and scientific research strength of West China Hospital of Sichuan University and the policy and platform advantages of Boao Lecheng, focusing on innovative drugs And medical device approval and post-market supervision, build a real-world data platform of "Lecheng-Hainan-Nationwide".
Based on the overall construction idea of "based on Lecheng, radiating Hainan" to "based on Hainan, radiating the whole country", the laboratory takes Boao Lecheng as an experimental field to innovate real-world data research practices, and form a series of research results with the characteristics of Hainan Free Trade Port. In the future, it will play an important role in building a domestic leading and internationally advanced real-world data research system and cultivating and exporting high-level, multi-disciplinary talent echelons, and will provide powerful technologies for my country to seize the "world first" in the field of clinical real-world data application. support.